Description
Suitable for electrophoresis, ≥99%
Synonyms: 2-Propenamide, Polyacrylamide, acrylic amide, akrylamid, Acrylic acid amide
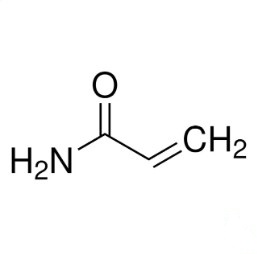
Linear Formula: CH2=CHCONH2
Molecular Weight: 71.08
General description
Acrylamide acts as a quencher of tryptophan fluorescence. Acrylamide alkylates SH groups of cysteine and ε-NH2 lysine residues of proteins during electrophoresis. Bis-acrylamide is required for crosslinking and acrylamide is vital for the polymerization of monomers. Acrylamide along with bis-acrylamide crosslinks to form polyacrylamide gels, which is used to separate protein and other biomolecules.
Acrylamide (AA) is a neurotoxin and carcinogen. Potato products like, French fries, potato chips and also several cereal-based products contains acrylamide. This highly reactive organic compound has the ability to polymerize to form polyacrylamide.
Application
Acrylamide has been used:
- for the synthesis of hydrogels
- for the preparation of polyacrylamide gel
- to agglomerate vimentin and keratin intermediate filament network
Acrylamide has been used:
- to agglomerate the vimentin and keratin intermediate filament network of K562cells
- in the preparation and characterization of soft culture substrate[1]
- in hydrogel synthesis
- for determination of protein by sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE) electrophoresis
Properties
vapor density
2.45 (vs air)
Pack Size
500g
vapor pressure
0.03 mmHg ( 40 °C)
assay
≥99%
form
crystals
technique(s)
electrophoresis: suitable
bp
125 °C/25 mmHg (lit.)
mp
82-86 °C (lit.)
solubility
water: soluble 2.5 g/10 mL, clear, colorless
suitability
suitable for electrophoresis




Reviews
There are no reviews yet.